Most of the material presented here has been published (Lensink, C. Tetrahedron: Asymmetry, 1995, 6, 2033-2038.) What follows is a summary of the ligand synthesis so it can be put into perspective with respect to the early transition metal complex preparation described in the other parts of this poster.
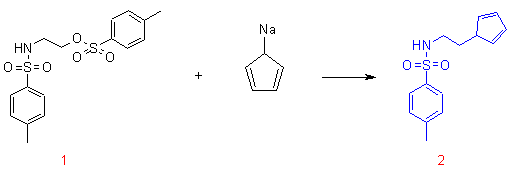
1,2-amino alcohols such as ethanol amine can be readily tosylated to yield the bis-tosylate starting material 1. Compound 1 reacts with an excess of sodium cyclopentadienide in THF to yield ligand 2 in high yield as indicated in the scheme below.

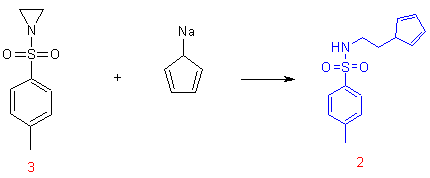
This reaction proceeds through N-tosylate aziridine intermediate and therefor, the N-tosylate aziridine 3 can also be used as the starting material in a stoichiometric reaction with sodium-cyclopentadienide as indicated below.
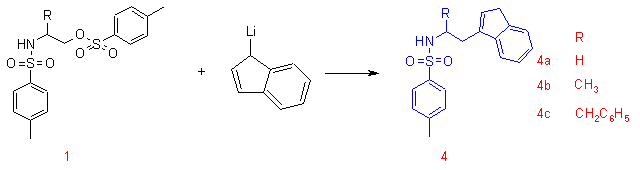
Lithium indenide reacts readily with a number of tosylated amino alcohols as well to yield the ligands 4a-c. The synthetic methodology can be extended to the synthesis of chiral, enantiomerically pure ligands by starting from chiral amino alcohols. Examples of these chrial ligands are 4b and 4c.
A single crystal structure determination was carried out for ligand 4a.
Experimental details and nmr data are given below.
2: To a solution of NaC5H5 (37 mmol, prepared from NaH and C5H6) in THF (20 mL) was added dropwise a solution of 1 (4.0 g, 20.3 mmol) in THF (10 mL). The reaction mixture was stirred overnight at room temperature. A saturated solution of NH4Cl (20 mL) was added to the reaction mixture. The organic phase was separated from the aqueous phase. The aqueous phase was extracted with ethyl acetate. The combined organic phases were concentrated under reduced pressure to yield a brown oil (2.34 g) The oil was subjected to column chromatography (silica-60, ethyl acetate/hexanes 1/3). Compound 2 was isolated as a mixture of two isomers as a pale yellow oil which crystallized on standing (2.60 g, 48 %). (Found; C, 63.20; H, 6.51; N, 5.39; S, 11.80. C14H17NO2S requires C, 63.85; H, 6.51; N, 5.32; S, 12.17). dH (CDCl3) 2.35 (s, 3H, CH3), 2.48 (m, 2H, NCH2CH2), 2.68/2.85 (both d, 2H, J 1.3 Hz, CH2(indene)), 3.05 (m, 2H, NCH2), 4.54 (br, 1H, NH), 5.93(s), 6.07(s), 6.21(m) and 6.30(m) (these 4 signals integrate for 3H, CH of the Cp ring), 7.22 (d, 2H, J 8.1 Hz, CHarom(tosyl)), 7.65 (d, 2H, J 8.3 Hz, CHarom(tosyl)); dC (CDCl3) 21.4 (CH3), 29.6, 30.4 (CH2(indene)), 41.4, 41.9 (NCH2CH2), 42.6, 42.9 (NCH2), 127.0, 128.5, 128.9, 131.7, 132.1, 133.5, 134.7, 136.8, 136.9, 142.5, 143.3, 144.3.
4a: Freshly distilled indene (3.74 g, 32.6 mmol) was dissolved in dry THF (100 mL) and cooled to -78 oC. nBuLi (13 mL, 1.6 M) was added via syringe. The reaction mixture was allowed to warm to 0 oC. The reaction mixture was subsequently cooled to -40 oC and 1 (4.0 g, 20.3 mmol) was added as a solid in one portion. The reaction mixture was allowed to warm slowly to room temperature and stirred for 17h. Saturated NH4Cl (100 mL) was added to the reaction mixture followed by ethyl acetate (50 mL). The organic layer was separated from the aqueous layer, washed with brine and dried on Na2SO4. The solvent was removed in vacuoto yield a tan coloured solid. Recrystallization from hexanes/ethyl acetate (2/1) yielded 4a as off-white crystals (4.06 g, 64 %). (Found; C, 68.81; H, 6.11; N, 4.47; S, 10.21. C18H19NO2S requires C, 68.98; H, 6.11; N, 4.47; S, 10.23 %). dH (CDCl3) 2.31 (3H, s, CH3), 2.64 (2H, dt, J 1.4 Hz and 6.8 Hz, NCH2CH2), 3.20 (q, 2H, J 6.8 Hz, NCH2), 3.21 (s, 2H, CH2(indene)), 4.60 (t, 1H, J 6.0 Hz, NH), 6.11 (s, 1H, CH(indene)) 7.12 (m, 3H, CHarom(indene)), 7.14 (d, 2H, J 8.0 Hz, CHarom(tosyl)), 7.35 (m, 1H, CHarom(indene)), 7.61 (d, 2H, J 8.0 Hz, CHarom(tosyl)); dC (CDCl3) 21.5 (CH3), 28.0 (CH2(indene)), 37.9 (NCH2CH2), 41.4 (NCH2CH2), 118.7, 123.9, 124.90, 126.1, 127.0, 129.6, 130.2, 136.9, 140.2, 143.3, 144.3, 144.3.
Alternatively 4a can be prepared from LiC9H7 (21.3 mmol) and 3 (2.50 g, 6.77 mmol). Yield 1.61 g, 76 %. The product had 1H and 13C spectra identical to the product described in the previous procedure.
4b: From LiC9H7 (20 mmol) and 1b (1.65 g, 4.30 mmol) prepared by tosylation of (R)-(-)-2-amino-1-propanol, chromatography (silica-60, ethyl acetate/hexanes 2/5). The product was isolated a pale brown oil (1.24 g, 88 %). [alpha]D: -32.1 (c = 0.42, CHCl3); dH (CDCl3) 1.14 (d, 3H, J 6.4 Hz, CHCH3), 2.26 (s, 3H, C6H5CH3), 2.55 (dd, 1H, 2J = 14.1 Hz, 3J 6.6 Hz, NCHCHa), 2.61 (dd, 1H, 2J = 14.1 Hz, 3J = 7.1 Hz, NCHCHb), 3.16 (s, 2H, CH2(indene)), 3.51 (m, 1H, NCH), 4.38 (d, 1H, J 6.3 Hz, NH), 6.10 (s, 1H, CH(indene)), 6.98-7.60 (m, 8H). dC (CDCl3) 21.4 (C6H5CH3), 22.1 (NCHCH3), 35.9 (NCHCH2), 37.8 (CH2(indene)), 48.4 (NCH), 118.9, 123.7, 124.6, 126.0, 126.7, 129.3, 131.2, 137.1, 140.0, 142.9, 144.2.
4c: From LiC9H7 (20 mmol) and 2c (1.70 g, 3.98 mmol) prepared by tosylation of (S)-(-)-2-amino-3-phenyl-1-propanol, chromatography (silica-60, ethyl acetate/hexanes 1/3). The product was further purified by recrystallization from warm ethyl acetate/hexanes (1/10) (1.34g, 79 %). [alpha]D: +23.3 (c = 0.37, CHCl3); (Found; C, 74.38; H, 6.42; N, 3.72; S, 7.95. C25H25NO2S requires C, 74.41; H, 6.24; N, 3.47; S, 7.94). dH (CDCl3) 2.21 (s, 3H, CH3), 2.57 (dd, 1H, 2J = 14.4 Hz, 3J = 7.9 Hz, NCHCHa), 2.65 (dd, 1H, 2J = 14.4 Hz, 3J = 5.9 Hz, NCHCHb), 2.81 (dd, 1H, 2J = 13.7 Hz, 3J 6.9 Hz, C6H5CHa), 2.89 (dd, 1H, 2J 13.7 Hz, 3J 5.7 Hz, C6H5CHb), 3.11 (d, 2H, J 6.5 Hz, 2H, CH2(indene)), 3.58 (m, 1H, NCH), 4.35 (br, 1H, NH), 6.09 (s, 1H, CH(indene)), 6.84-7.41 (m, 13H); dC (CDCl3) 21.5 (CH3), 33.2 (C6H5CH2), 37.9 (CH2(indene)), 41.8 (NCHCH2), 53.7 (NCH), 119.1, 123.8, 124.8, 12 6.2, 126.7, 128.7, 129.1, 129.3, 129.7, 131.4, 140.2, 142.9, 144.3.