In the first part of the post on this topic, I described how an asymmetric sulfoxide could be prepared as a pure enantiomer using a chiral oxygen transfer reagent. In the second part, we now need to deliver a different group, cyano, to a specific face of the previously prepared sulfoxide-imine. The sulfoxide is now acting as a chiral auxilliary, and helps direct the delivery of the cyanide group to specifically one face of the imine rather than the other. After removal of the aluminum carrier for the cyano group and hydrolysis of the cyano group to a carboxylic acid group, we end up with an enantiomerically pure amino acid.
Archive for the ‘Interesting chemistry’ Category
Anatomy of an asymmetric reaction. The Strecker synthesis, part 2.
Wednesday, May 26th, 2010Anatomy of an asymmetric reaction. The Strecker synthesis, part 1.
Monday, May 24th, 2010The assembly of a molecule for a purpose has developed into an art form, one arguably (chemists always argue) that is approaching its 100th birthday (DOI: 10.1002/cber.191104403216) celebrating Willstätter’s report of the synthesis of cyclo-octatetraene. Most would agree it reached its most famous achievement with Woodward’s synthesis of quinine (DOI: 10.1021/ja01221a051) in 1944. To start with, the art was in knowing how and in which order to join up all the bonds of a target. The first synthesis in which (relative) stereocontrol of those bonds was the primary objective was reported in 1951 (10.1021/ja01098a039). The art can be taken one step further. It involves control of the absolute stereochemistry, involving making one enantiomer specifically (rather than the mirror image, which of course has the same relative stereochemistry). Nowadays, a synthesis is considered flawed if the enantiomeric excess (of the desired vs the undesired isomer) of such a synthesis does not achieve at least ~98%. It is routine. But ask the people who design such syntheses if they know exactly the reasons why their reaction has succeeded, you may get a less precise answer (or just a lot of handwaving; chemists also like to wave their hands as well as argue).
A Digital chemical repository – is it being used?
Tuesday, May 4th, 2010In this previous blog post I wrote about one way in which we have enhanced the journal article. Associated with that enhancement, and also sprinkled liberally throughout this blog, are links to a Digital Repository (if you want to read all about it, see DOI: 10.1021/ci7004737). It is a fairly specific repository for chemistry, with about 5000 entries. These are mostly the results of quantum mechanical calculations on molecules (together with a much smaller number of spectra, crystal structure and general document depositions). Today, with some help (thanks Matt!), I decided to take a look at how much use the repository was receiving.
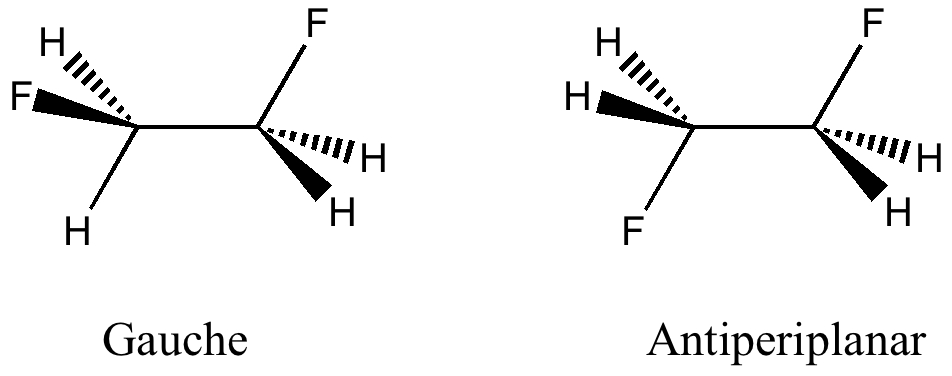
The conformation of 1,2-difluoroethane
Tuesday, April 6th, 2010Here I offer another spin-off from writing a lecture course on conformational analysis. This is the famous example of why 1,2-difluoroethane adopts a gauche rather than antiperiplanar conformation.
The structure of the hydrogen ion in water.
Sunday, February 21st, 2010Stoyanov, Stoyanova and Reed recently published on the structure of the hydrogen ion in water. Their model was H(H2O)n+, where n=6 (DOI: 10.1021/ja9101826). This suggestion was picked up by Steve Bachrach on his blog, where he added a further three structures to the proposed list, and noted of course that with this type of system there must be a fair chance that the true structure consists of a well-distributed Boltzmann population of a number of almost iso-energetic forms.
Conformational analysis of cyclotriborazane
Sunday, February 14th, 2010In an earlier post, I re-visited the conformational analysis of cyclohexane by looking at the vibrations of the entirely planar form (of D6h symmetry). The method also gave interesting results for the larger cyclo-octane ring. How about a larger leap into the unknown?
The conformation of cyclohexane
Thursday, January 28th, 2010Like benzene, its fully saturated version cyclohexane represents an icon of organic chemistry. By 1890, the structure of planar benzene was pretty much understood, but organic chemistry was still struggling somewhat to fully embrace three rather than two dimensions. A grand-old-man of organic chemistry at the time, Adolf von Baeyer, believed that cyclohexane too was flat, and what he said went. So when a young upstart named Hermann Sachse suggested it was not flat, and furthermore could exist in two forms, which we now call chair and boat, no-one believed him. His was a trigonometric proof, deriving from the tetrahedral angle of 109.47 at carbon, and producing what he termed strainless rings.