Archive for the ‘Interesting chemistry’ Category
Thursday, March 24th, 2022
The meeting covered the scientific life of Professor Sir Geoffrey Wilkinson from the perspective of collaborators, friends and family and celebrated three anniversaries, the centenary of his birth (2021), the half-century anniversary of the Nobel prize (2023) and 70 years almost to the day (1 April) since the publication of the seminal article on Ferrocene (2022).[1]
(more…)
References
- G. Wilkinson, M. Rosenblum, M.C. Whiting, and R.B. Woodward, "THE STRUCTURE OF IRON BIS-CYCLOPENTADIENYL", Journal of the American Chemical Society, vol. 74, pp. 2125-2126, 1952. https://doi.org/10.1021/ja01128a527
Posted in Historical, Interesting chemistry | No Comments »
Tuesday, March 22nd, 2022
Normally, aromaticity is qualitatively assessed using an electron counting rule for cyclic conjugated rings. The best known is the Hückel 4n+2 rule (n=0,1, etc) for inferring diatropic aromatic ring currents in singlet-state π-conjugated cyclic molecules‡ and a counter 4n rule which infers an antiaromatic paratropic ring current for the system. Some complex rings can sustain both types of ring currents in concentric rings or regions within the molecule, i.e. both diatropic and paratropic regions. Open shell (triplet state) molecules have their own rule; this time the molecule has a diatropic ring current if it follows a 4n rule, often called Baird’s rule. But has a molecule which simultaneously follows both Hückel’s AND Baird’s rule ever been suggested? Well, here is one, as indeed I promised in the previous post.
(more…)
Posted in Interesting chemistry | 2 Comments »
Saturday, March 19th, 2022
I discussed in the previous post the small molecule C4 and how of the sixteen valence electrons, eight were left over after forming C-C σ-bonds which partitioned into six σ and two π. So now to consider B4. This has four electrons less, and now the partitioning is two σ and two π (CCSD(T)/Def2-TZVPPD calculation, FAIR DOI: 10.14469/hpc/10157). Again both these sets fit the Hückel 4n+2 rule (n=0).
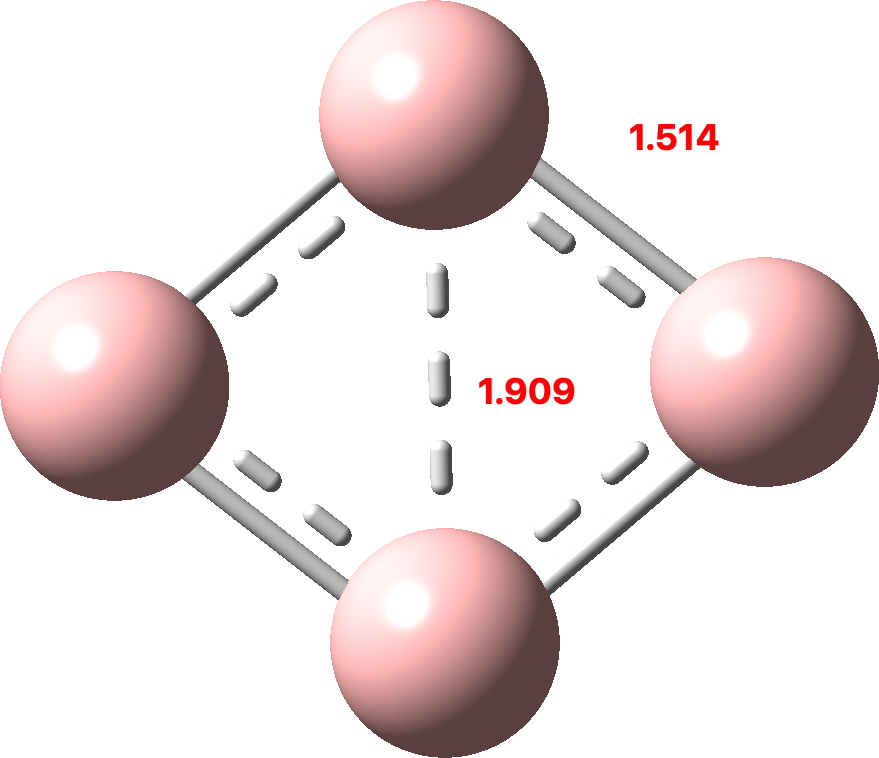 (more…)
(more…)
Posted in Interesting chemistry | No Comments »
Thursday, December 16th, 2021
The annual “molecule of the year” results for 2021 are now available … and the winner is Infinitene.[1],[2] This is a benzocirculene in the form of a figure eight loop (the infinity symbol), a shape which is also called a lemniscate [3] after the mathematical (2D) function due to Bernoulli. The most common class of molecule which exhibits this (well known) motif are hexaphyrins (hexaporphyrins; porphyrin is a tetraphyrin)[4],[5],[6], many of which exhibit lemniscular topology as determined from a crystal structure. Straightforward annulenes have also been noted to display this[7] (as first suggested here for a [14]annulene[8]) and other molecules show higher-order Möbius forms such as trefoil knots.[9],[10] This new example uses twelve benzo groups instead of six porphyrin units to construct the lemniscate. So the motif is not new, but this is the first time it has been constructed purely from benzene rings. (more…)
References
- K. Itami, M. Krzeszewski, and H. Ito, "Infinitene: A Helically Twisted Figure-Eight [12]Circulene Topoisomer", 2021. https://doi.org/10.26434/chemrxiv-2021-pcwcc
- M. Krzeszewski, H. Ito, and K. Itami, "Infinitene: A Helically Twisted Figure-Eight [12]Circulene Topoisomer", Journal of the American Chemical Society, vol. 144, pp. 862-871, 2021. https://doi.org/10.1021/jacs.1c10807
- C.S.M. Allan, and H.S. Rzepa, "Chiral Aromaticities. AIM and ELF Critical Point and NICS Magnetic Analyses of Möbius-Type Aromaticity and Homoaromaticity in Lemniscular Annulenes and Hexaphyrins", The Journal of Organic Chemistry, vol. 73, pp. 6615-6622, 2008. https://doi.org/10.1021/jo801022b
- H. Rath, J. Sankar, V. PrabhuRaja, T.K. ChandrashekarPresent address: The D, B.S. Joshi, and R. Roy, "Figure-eight aromatic core-modified octaphyrins with six meso links: syntheses and structural characterization", Chemical Communications, pp. 3343, 2005. https://doi.org/10.1039/b502327k
- H. Rath, J. Sankar, V. PrabhuRaja, T.K. Chandrashekar, and B.S. Joshi, "Aromatic Core-Modified Twisted Heptaphyrins[1.1.1.1.1.1.0]: Syntheses and Structural Characterization", Organic Letters, vol. 7, pp. 5445-5448, 2005. https://doi.org/10.1021/ol0521937
- S. Shimizu, N. Aratani, and A. Osuka, "<i>meso</i>‐Trifluoromethyl‐Substituted Expanded Porphyrins", Chemistry – A European Journal, vol. 12, pp. 4909-4918, 2006. https://doi.org/10.1002/chem.200600158
- T. Perera, F.R. Fronczek, and S.F. Watkins, "2,9,16,23-Tetrakis(1-methylethyl)-5,6,11,12,13,14,19,20,25,26,27,28-dodecadehydrotetrabenzo[<i>a</i>,<i>e</i>,<i>k</i>,<i>o</i>]cycloeicosene", Acta Crystallographica Section E Structure Reports Online, vol. 67, pp. o3493-o3493, 2011. https://doi.org/10.1107/s1600536811048604
- H.S. Rzepa, "A Double-Twist Möbius-Aromatic Conformation of [14]Annulene", Organic Letters, vol. 7, pp. 4637-4639, 2005. https://doi.org/10.1021/ol0518333
- G.R. Schaller, F. Topić, K. Rissanen, Y. Okamoto, J. Shen, and R. Herges, "Design and synthesis of the first triply twisted Möbius annulene", Nature Chemistry, vol. 6, pp. 608-613, 2014. https://doi.org/10.1038/nchem.1955
- S.M. Bachrach, and H.S. Rzepa, "Cycloparaphenylene Möbius trefoils", Chemical Communications, vol. 56, pp. 13567-13570, 2020. https://doi.org/10.1039/d0cc04190d
Posted in Chiroptics, Interesting chemistry | 5 Comments »
Saturday, November 27th, 2021
The title comes from the abstract of an article[1] analysing why Biotin (vitamin B7) is such a strong and effective binder to proteins, with a free energy of (non-covalent) binding approaching 21 kcal/mol. The author argues that an accumulation of both CH-π and CH-O together with more classical hydrogen bonds and augmented by a sulfur centered hydrogen bond, oxyanion holes and water solvation, accounts for this large binding energy. 
(more…)
References
- D.B. McConnell, "Biotin’s Lessons in Drug Design", Journal of Medicinal Chemistry, vol. 64, pp. 16319-16327, 2021. https://doi.org/10.1021/acs.jmedchem.1c00975
Posted in Interesting chemistry | No Comments »
Thursday, October 14th, 2021
On October 13, 2021, the historical group of the Royal Society of Chemistry organised a symposium celebrating ~150 years of the history of (molecular) chirality. We met for the first time in person for more than 18 months and were treated to a splendid and diverse program about the subject. The first speaker was Professor John Steeds from Bristol, talking about the early history of light and the discovery of its polarisation. When a slide was shown about herapathite[1] my “antennae” started vibrating. This is a crystalline substance made by combining elemental iodine with quinine in acidic conditions and was first discovered by William Herapath as long ago as 1852[2] in unusual circumstances. Now to the serendipity!
(more…)
References
- B. Kahr, J. Freudenthal, S. Phillips, and W. Kaminsky, "Herapathite", Science, vol. 324, pp. 1407-1407, 2009. https://doi.org/10.1126/science.1173605
- W.B. Herapath, "XXVI. <i>On the optical properties of a newly-discovered salt of quinine, which crystalline substance possesses the power of polarizing a ray of light, like tourmaline, and at certain angles of rotation of depolarizing it, like selenite</i>", The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science, vol. 3, pp. 161-173, 1852. https://doi.org/10.1080/14786445208646983
Posted in Chiroptics, crystal_structure_mining, Historical, Interesting chemistry | No Comments »
Tuesday, August 17th, 2021
The homologous hydrocarbon series R4C is known for R=Me as neopentane and for R=Et as 3,3-diethylpentane. The next homologue, R=iPr bis(3,3-isopropyl)-2,4-dimethylpentane is also a known molecule[1] for which a crystal structure has been reported (DOI: https://doi.org/10.5517/cc4wvnh). The final member of the series, R= tbutyl is unknown. Here I have a look at some properties of the last two of these highly hindered hydrocarbons.
(more…)
References
- S.I. Kozhushkov, R.R. Kostikov, A.P. Molchanov, R. Boese, J. Benet-Buchholz, P.R. Schreiner, C. Rinderspacher, I. Ghiviriga, and A. de Meijere, "Tetracyclopropylmethane: A Unique Hydrocarbon with S4 Symmetry", Angewandte Chemie International Edition, vol. 40, pp. 180-183, 2001. https://doi.org/10.1002/1521-3773(20010105)40:1<180::aid-anie180>3.0.co;2-k
Posted in Interesting chemistry | No Comments »
