June 6th, 2014
Following the discussion here of Kekulé’s suggestion of what we now call a vibrational mode (and which in fact now bears his name), I thought I might apply the concept to a recent molecule known as [2.2]paracyclophane. The idea was sparked by Steve Bachrach’s latest post, where the “zero-point” structure of the molecule has recently been clarified as having D2 symmetry.[1]
Read the rest of this entry »
References
- H. Wolf, D. Leusser, M. Jørgensen, R. Herbst‐Irmer, Y. Chen, E. Scheidt, W. Scherer, B.B. Iversen, and D. Stalke, "Phase Transition of [2,2]‐Paracyclophane – An End to an Apparently Endless Story", Chemistry – A European Journal, vol. 20, pp. 7048-7053, 2014. https://doi.org/10.1002/chem.201304972
Tags: 10.1002, 201304972, Hiberty and co, Steve Bachrach
Posted in Historical, Interesting chemistry | No Comments »
May 28th, 2014
In the preceding post, a nice discussion broke out about Kekulé’s 1872 model for benzene.[1] This model has become known as the oscillation hypothesis between two extreme forms of benzene (below). The discussion centered around the semantics of the term oscillation compared to vibration (a synonym or not?) and the timescale implied by each word. The original article is in german, but more significantly, obtainable only with difficulty. Thus I cannot access[1] the article directly since my university does not have the appropriate “back-number” subscription.‡ So it was with delight that I tracked down an English translation in a journal that I could easily access.[2] Here I discuss what I found (on pages 614-615, the translation does not have its own DOI).
Read the rest of this entry »
References
- A. Kekulé, "Ueber einige Condensationsproducte des Aldehyds", Justus Liebigs Annalen der Chemie, vol. 162, pp. 77-124, 1872. https://doi.org/10.1002/jlac.18721620110
- "Organic chemistry", Journal of the Chemical Society, vol. 25, pp. 605, 1872. https://doi.org/10.1039/js8722500605
Tags: Henry Armstrong, Kekule vibration, Paul Schleyer
Posted in Historical | 10 Comments »
May 18th, 2014
Continuing my european visits, here are two photos from Bonn. First, a word about how the representation of benzene evolved, attributed to Kekulé.
Read the rest of this entry »
Tags: /RT, free energy barrier, gas constant
Posted in Historical | 3 Comments »
May 18th, 2014
Not a computer in sight! I refer to a chemistry lab from the 1800s I was recently taken to, where famous french chemists such as Joseph Gay-Lussac, Michel Chevreul and Edmond Fremy were professors. Although not used for chemistry any more, it is an incredible treasure trove of objects. Here are photos of some.
Read the rest of this entry »
Tags: Edmond Fremy, Joseph Gay-Lussac, Michel Chevreul
Posted in Historical | No Comments »
May 17th, 2014
I remember a time when tracking down a particular property of a specified molecule was an all day effort, spent in the central library (or further afield). Then came the likes of STN Online (~1980) and later Beilstein. But only if your institution had a subscription. Let me then cut to the chase: consider this URL: http://search.datacite.org/ui?q=InChIKey%3DLQPOSWKBQVCBKS-PGMHMLKASA-N The site is datacite, which collects metadata about cited data! Most of that data is open in the sense that it can be retrieved without a subscription (but see here that it is not always made easy to do so). So, the above is a search for cited data which contains the InChIkey LQPOSWKBQVCBKS-PGMHMLKASA-N. This produces the result:
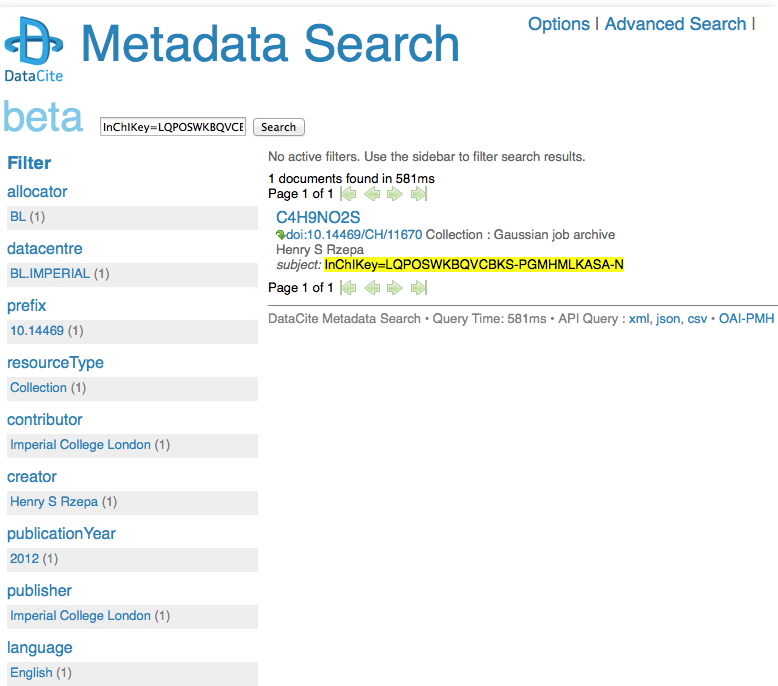
This tells you who published the data (but oddly, its date is merely to the nearest year? It is beta software after all). The advanced equivalent of this search looks like this:
Read the rest of this entry »
Tags: beta software, generic data search, molecular search engines, search engine, search engine optimisation, search looks, software agents
Posted in Chemical IT | No Comments »
May 5th, 2014
My name is displayed pretty prominently on this blog, but it is not always easy to find out who the real person is behind many a blog. In science, I am troubled by such anonymity. Well, a new era is about to hit us. When you come across an Internet resource, or an opinion/review of some scientific topic, I argue here that you should immediately ask: “what is its provenance?”
Read the rest of this entry »
Tags: 0000-0002-8635-8390, added value site, API, Internet resource, ORCiD, programmer, United Kingdom
Posted in Chemical IT | No Comments »
May 2nd, 2014
This is rather cranking the handle, but taking my previous post and altering the search definition of the crystal structure database from 4- to 5-coordinate metals, one gets the following.
Read the rest of this entry »
Tags: Fe and Co, search definition
Posted in Chemical IT, crystal_structure_mining, General | 1 Comment »
April 30th, 2014
I love experiments where the insight-to-time-taken ratio is high. This one pertains to exploring the coordination chemistry of the transition metal region of the periodic table; specifically the tetra-coordination of the series headed by Mn-Ni. Is the geometry tetrahedral, square planar, or other? One can get a statistical answer in about ten minutes.
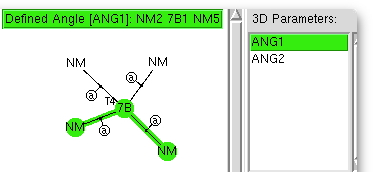 The (CCDC database) search definition required is shown above. The central atom defines the column of the period table, it is specified to have precisely four other atoms bonded to it, which can be any other element. These four bonds are specified as acyclic (to avoid any bias introduced by rings). And two angles are defined subtending the central atom. And off we go, defining on the way that the hits must be refined to an R-factor of < 0.05, have no disorder, and no errors.
The (CCDC database) search definition required is shown above. The central atom defines the column of the period table, it is specified to have precisely four other atoms bonded to it, which can be any other element. These four bonds are specified as acyclic (to avoid any bias introduced by rings). And two angles are defined subtending the central atom. And off we go, defining on the way that the hits must be refined to an R-factor of < 0.05, have no disorder, and no errors.
Read the rest of this entry »
Tags: data, Pt[/caption] Square, search definition, transition metal region
Posted in Chemical IT, crystal_structure_mining, General | No Comments »
April 20th, 2014
Ribulose-1,5-bisphosphate reacts with carbon dioxide to produce 3-keto-2-carboxyarabinitol 1,5-bisphosphate as the first step in the biochemical process of carbon fixation. It needs an enzyme to do this (Ribulose-1,5-bisphosphate carboxylase/oxygenase, or RuBisCO) and lots of ATP (adenosine triphosphate, produced by photosynthesis). Here I ask what the nature of the uncatalysed transition state is, and hence the task that might be facing the catalyst in reducing the activation barrier to that of a facile thermal reaction. I present my process in the order it was done‡.
Read the rest of this entry »
Tags: 1M solutions, carbon fixation, chair, chemist, energy, free energy, low energy, low energy sink, lower energy conformation, lower energy isomer, Peter Medawar, phosphate
Posted in Interesting chemistry, reaction mechanism | 1 Comment »

