Chemists now use the term “curly arrows” as a language to describe the electronic rearrangements that occur when a (predominately organic) molecule transforms to another – the so called chemical reaction. It is also used to infer, via valence bond or resonance theory, what the mechanistic implications of that reaction are. It was in this latter context that the very first such usage occured in 1924[1] taking the form of a letter by Robert Robinson to the secretary of the Chemical Society and “read” on December 18th 1924. The following diagram was included:
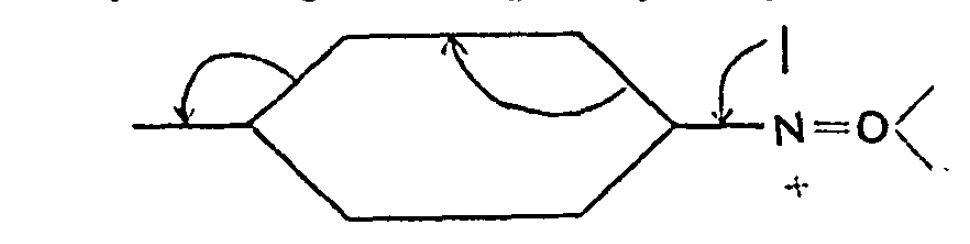
First curly arrows
I have commented previously on this diagram[cite10.59350/qqwk3-dgj13[/cite] and will not discuss it further here. To commemorate the 100th anniversary of their invention, I include shots of two “modern” sets of curly arrows, taken from a lecture I give to university students at the end of their first university year.
- The first was a new take on the peracid epoxidation of an alkene[2] in which quantum mechanical calculations have revealed that the classic take on the curly arrow mechanism for this reaction can be split into two sets, five for the first stage of the reaction up to the transition state and two for the final stage
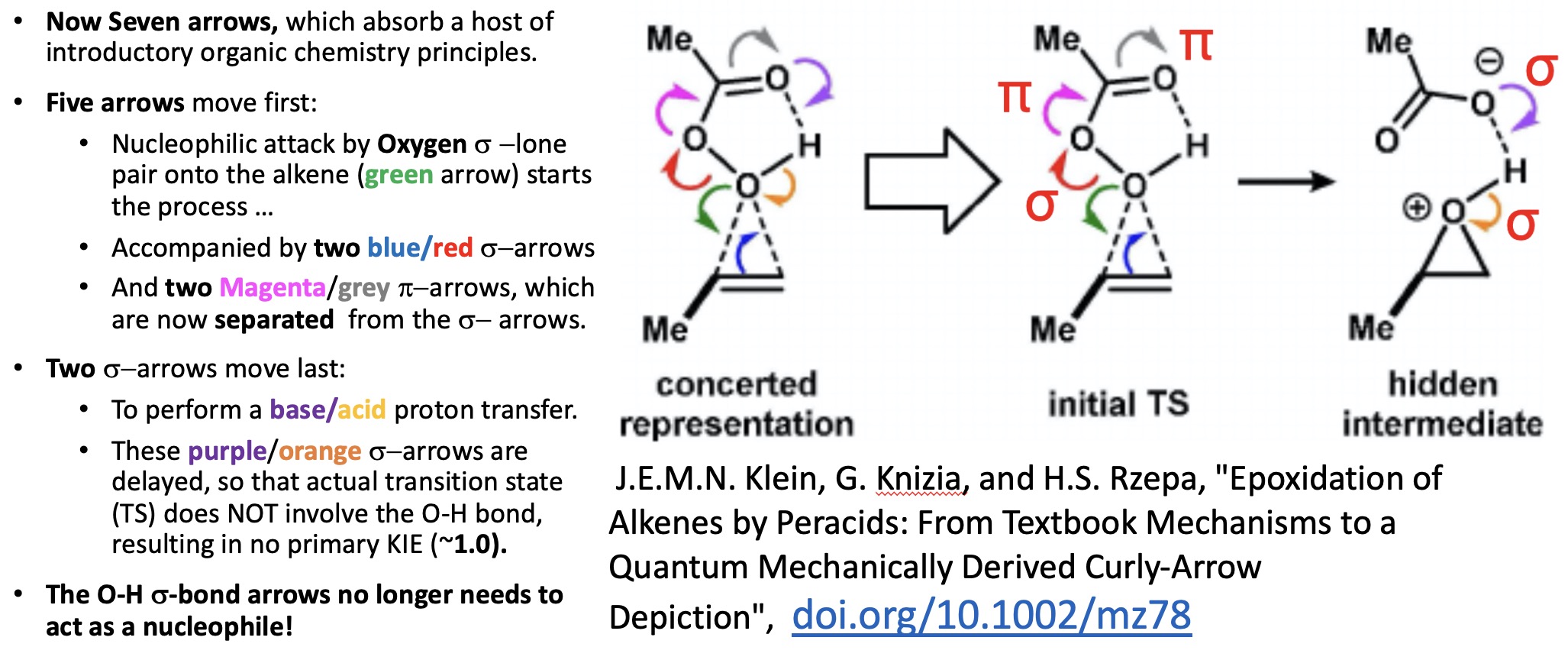
Four becomes seven
- The second was also discussed here[3] and involves what is arguably a new type of arrow to join the existing stable – the dashed arrow (in red below). This electron transfer arrow can take place over long distances (15Å or more) and adds the concept that an arrow can have the properties of an (approximate) length as well as direction, start+points and perhaps even “curlyness”.
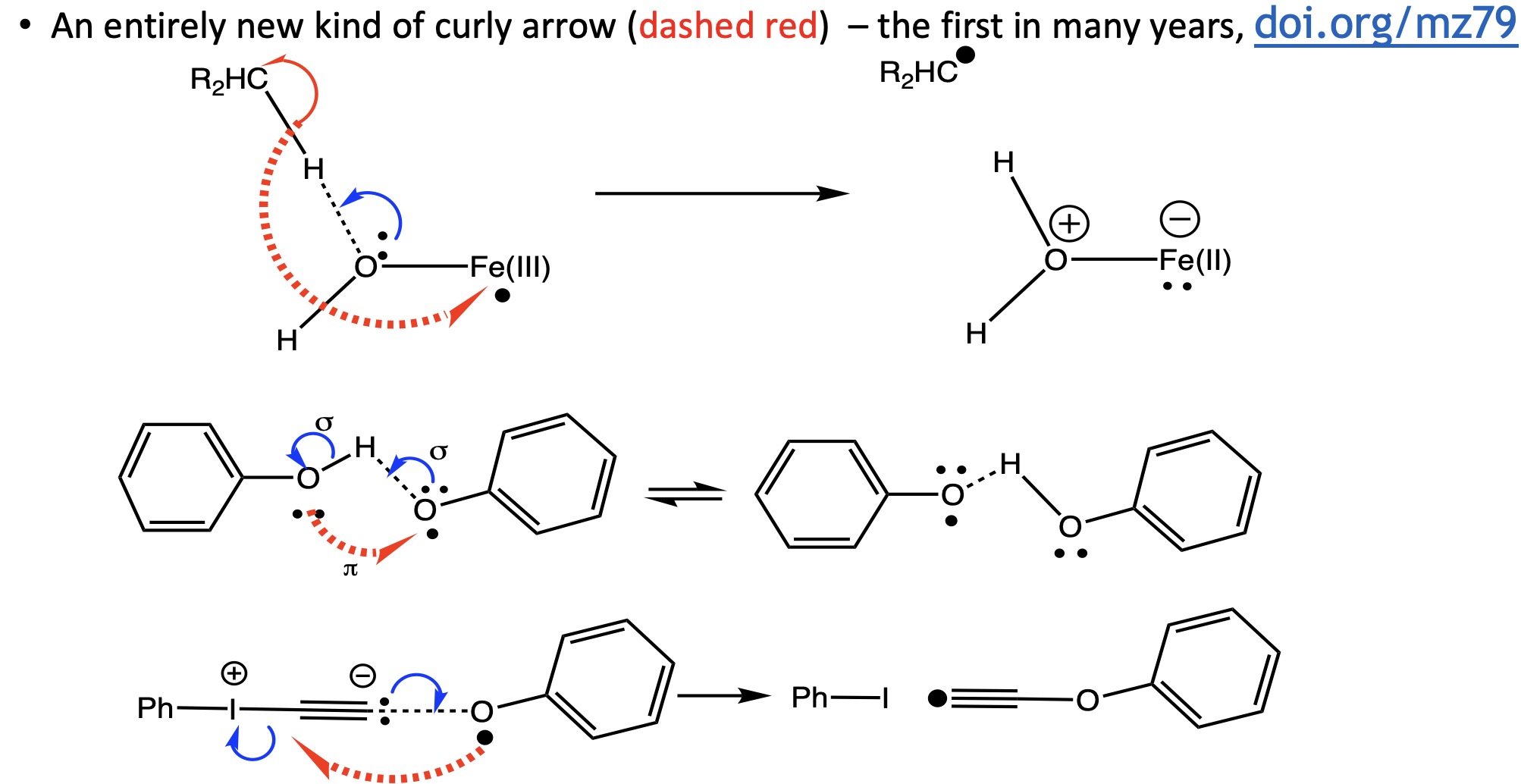
Proton coupled electron transfers
As a “language” describing mechanism and reactivity in molecules, curly arrows are still in common use, but as chemistry itself evolves into new areas, will curly arrows themselves morph into new forms, or will their use gradually decline?
References
- "Forthcoming events", Journal of the Society of Chemical Industry, vol. 43, pp. 1295-1298, 1924. https://doi.org/10.1002/jctb.5000435208
- H. Rzepa, "Experimental evidence for "hidden intermediates"? Epoxidation of ethene by peracid.", 2013. https://doi.org/10.59350/fdy9j-9fp48
- H. Rzepa, "Curly arrows in the 21st Century. Proton-coupled electron transfers.", 2020. https://doi.org/10.59350/rj90z-mxh96