Another common type of pericyclic reaction is the migration of hydrogen or carbon along a conjugated chain, as in the [1,3] migration of a carbon as shown below. As before, I explore the stereochemistry of the thermal and photochemical reactions.

The reaction is known to proceed thermally with inversion of configuration at the migrating carbon, and constitutes a 4n-electron reaction involving one antarafacial component (at that carbon). As usual, the thermal reaction can be rationalised by its transition state and an accompanying IRC.
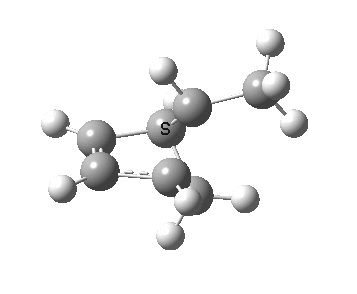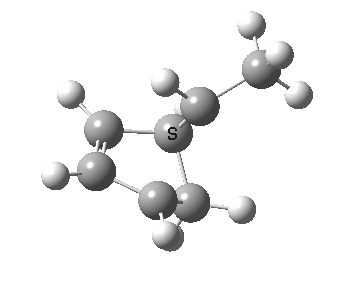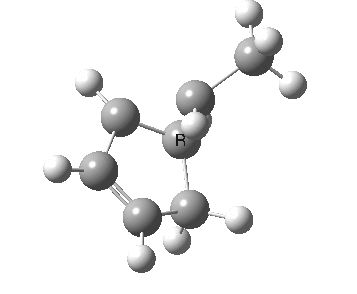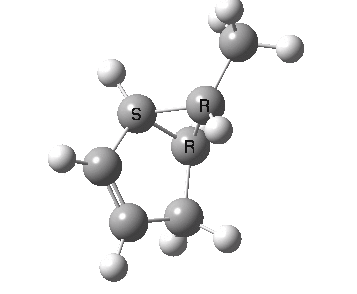
IRC for the thermal migration of a carbon. Click for 3D.
The photochemical reaction (not hitherto studied as far as I know) has a
conical intersection between the S
1 and S
0 states which clearly displays migration of the carbon with
retention (
suprafacially).
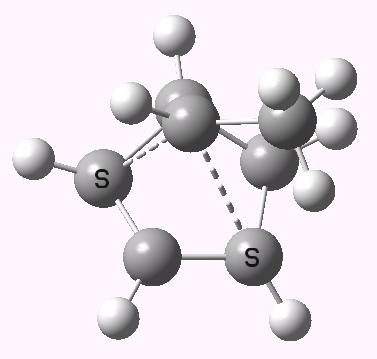
Photochemically induced 1,3 migration with retention of configuration. Click for 3D
I conclude this trio of pericyclic reactions with a re-affirmation of the general rules for such reactions, which state that changing the reaction conditions from heat to light is likely invert one suprafacial or antarafacial stereochemical component, located either on the transition state (for the thermal reaction) or the conical intersection (for the photochemical equivalent). A caveat is necessary, in that the pathways for photochemical reactions are generally more complex than thermal ones, and so exceptions to this broad statement may well be easy to find.
-
Henry Rzepa is Emeritus Professor of Computational Chemistry at Imperial College London.
View all posts
Related
Tags: pericyclic, Tutorial material
This entry was posted on Tuesday, November 29th, 2011 at 7:16 am and is filed under Uncategorised. You can follow any responses to this entry through the RSS 2.0 feed.
You can leave a response, or trackback from your own site.


[…] Henry Rzepa Chemistry with a twist « A modern take on pericyclic sigmatropic migrations. […]