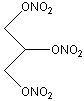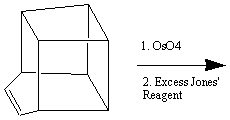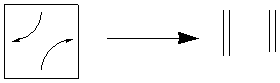Introduction
-
High Energy-Density Materials (HEDMs)
HEDMs are chemicals which have high enthalpies of formation. This means
that their reactions generate large amounts of energy, as the thermodynamically
unstable compound decomposes into more stable compounds. They are used for
several purposes:
- Explosives.
- Propellants.
- Fuels.
However, to be useful, an HEDM must have more then a high enthalpy
of formation. As well as being thermodynamically unstable, it must also
be kinetically stable, as it must be transportable, and storable - an HEDM
which decomposes five minutes after creation is no good to anyone! Also,
it helps if it is highly dense, as the higher the density, the higher the
energy density is, and the easier it is to transport.
Explosives and propellants have additional requirements. They must react
quickly under a fairly easily achieved condition, as their effectiveness
depends on their speed of reaction. This requires their being dense, as the
speed of the shock wave that causes detonation is proportional to the square
of the density of the explosive. The products of their reactions must also
have a much greater volume than the reactants - ideally, what is wanted is
a solid that decomposes to give multiple moles of gas. Because much
gas needs to be generated, many explosives and propellants are nitrogen and/or
oxygen rich. Some well-known HEDMs are shown below:
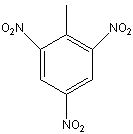
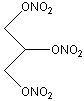
TNT
Nitroglycerin
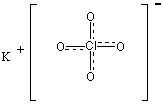
Potassium Chlorate (VII)
(1-methyl, 2, 4, 6-nitrobenzene)
(1, 2, 3-trinitratopropane)
Cubane
Cubane is a hydrocarbon with the molecular formula C8H8.
It, perhaps unsurprisingly, is formed with each carbon being at the vertex
of a cube. It had long been thought of as a potential HEDM before
anyone had ever synthesised it - indeed, many thought it would be so strained
that it would be impossible to make! With its very strained bond angles,
it can easily be seen why cubane's existence was thought impossible. However,
cubane was synthesised, and proved to be very stable at room temperature,
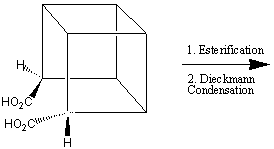
despite having an enthalpy of formation of +620 kJmol-11. One synthetic route2 for the synthesis of cubane is shown below:
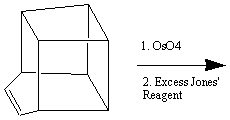
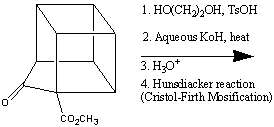
Cubane is surprisingly stable; it has a thermal decomposition energy of
180kJmol-11 in the gas phase at 230-260oC.
This is because there are no kinetically viable paths along which cubane
can decompose1. This can be seen quite
easily by comparing cubane to a similar molecule - cyclobutane. Cubane consists
of 6 cyclobutyl rings, so this is a fair comparison. The decomposition
can start to occur one of two ways - with one bond breaking, or with two bonds
breaking.
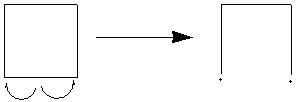
This decomposition route forms a diradical, an intermediate of high energy.
However, in cubane, this cyclobutyl ring will still be held in place by
the others, i.e. a hugely reactive intermediate will be formed, and no strain
will be released. Hence, this decomposition route cannot occur
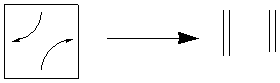
This reaction is a 2+2 cycloelimination, and is therefore symmetry forbidden
according to the Woodward-Hoffmann rules. This decomposition route cannot
occur.
However, cubane, like other hydrocarbons, does not react especially
quickly - it's a fairly good fuel (if you ignore its exorbitant price),
but it's not very useful as an explosive or a propellant. It also does not
have as high an enthalpy of formation as other modern HEDMs. However, like
other hydrocarbons, it can take other substituents, and these cubane derivatives
can potentially be much higher in energy than their parent molecule.